Consultancies in Microbiology, Anti-infectives, & Antimicrobial Resistance (AMR)
We have subject matter experts who have successfully delivered clinical candidates in anti-infectives from Microbiology, Molecular Biology, Medicinal Chemistry, Pharmacology, Drug metabolism and pharmacokinetics, Toxicology, Regulatory Affairs, Quality Assurance, and Clinical Trials.
Microbiology: Testing solutions (in Bacteriology, Mycology and virology), Clinical Microbiology, Qualification of products in Pharmaceutical, Nutraceutical, Medical Devices etc..
Clinical Microbiology:
Correlating of the isolate, and its antibiogram with the clinical history and advising on the right path of managing the patient, infection control practices, LIMS.
General Microbiology:
Interpretation of the data to correlate with the scientific question that is being pursued.
Clinical Trials :
Aligning the data and scientifically interpreting towards meaningful interpretation.
Institutional Ethics Committees:
Institutional Ethics Committees: Evaluation of proposals for human, and animal ethics in the light of research goals being pursued.
Institutional Biosafety Committees:
To evaluate and balance the risk, threat, and advantages of each microorganism that is being used for research purpose or as a tool for producing material of commercial value.
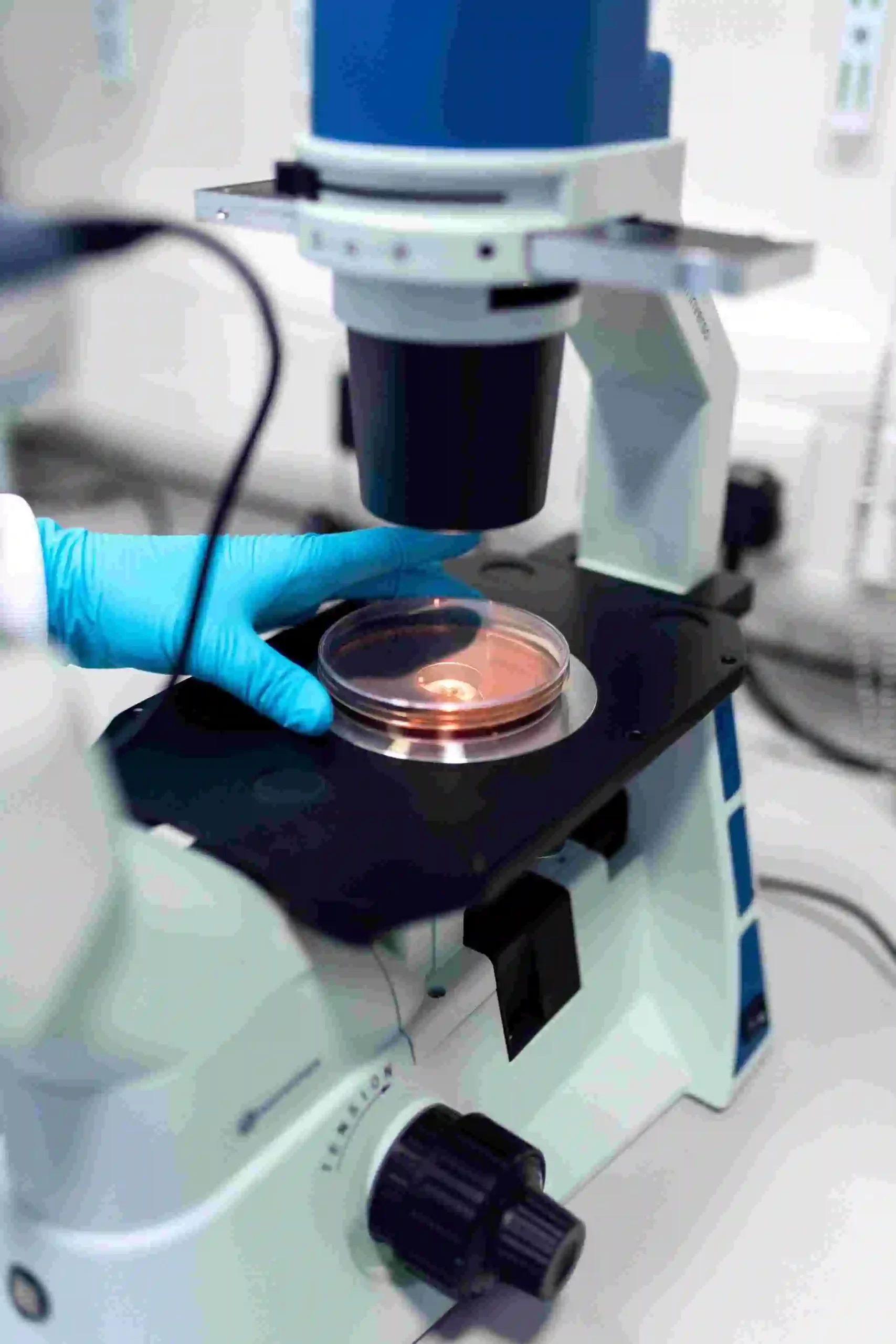
- Facilitate from decision-making to Testing of novel compounds / compound libraries for repurposing or combinations in anti-infectives.
- Interpretation of data to convert into information during the entire journey of a molecule from ideation up until Clinical trials.
Microvioma believes in the principle of Shared Solutions model maintaining strict industry standard confidentiality practices. Wet-lab facilitation for discovery of novel anti-infective compounds through our collaborative model. Some of the core investigations include:
- Mechanism of action (MoA) determination and in vitro target validation studies.
- Industry-standard methods adopted, incl. CLSI, to test compounds for antimicrobial activity against organisms esp. the ESKAPE.
- Screening for hit identification of antimicrobial activity on a BSL 2 contained platform.
- MIC, MBC/MFC, time-kill, PAE and intracellular killing studies using single or combination agents.
- Frequency of resistance determination and detailed characterisation of resistant mutants.
- Early assessment of cytotoxicity potential against multiple mammalian cell lines.
- Non-clinical in vitro and in vivo PK/PD infection models for the optimisation of drug concentration profiles to maximise bacterial killing and minimise the emergence of resistance.
- Compound/drug combination studies for assessment of synergistic, antagonistic and additive effects.
- Assessing the efficacy of lead and candidate compounds in highly relevant and validated disease models across all important pathogens including ESKAPE organisms, & fungi.

The discovery of antibiotics was the inflection point in human existence since it contributed enormously to the extension of the human lifespan. However, this excitement was tempered by the detection of antimicrobial resistance (AMR) prior to the launch of the first antibiotic (Penicillin), which continues to be a major deterrent leading to significant mortality and morbidity. The drivers of antimicrobial resistance include antimicrobial use and abuse in human, animal, and environmental sectors and the spread of resistant bacteria and resistance determinants within and between these sectors and around the globe. This phenomenon has consistently evolved, as evidenced by several reports such as: GLASS (2021), 2020 AMR Benchmark (WEF), the DRIVE-AB (2018), and the Scoping Report on AMR in India (2017). The economic burden due to AMR related mortality is estimated to exponentially rise to above 10 million deaths per year by 2050 costing an estimated $100 trillion.
AMR must be understood as a response to an indiscriminate exposure of microbes to antibiotics rather than a microbial attack on human health. The very fact that we are using antibiotics in various sectors such as poultry, cattle, agriculture, food industry, human health, in addition to release into the environment is a clear indication that we are exploiting them, irrationally. AMR by itself is a complex and interdisciplinary public-health challenge driven by multiple factors, especially the (ab)use of antimicrobials in all the areas of the World Health Organization’s One Health coverage. This continued evolution of AMR has also impeded United Nation’s Sustainable Development Goals set to serve as a global blueprint for a better, more equitable, more sustainable life on our planet.
Microvioma participates in this AMR mission:
- Surveillance for microorganisms in any given environment and to correlate the interplay between the elements of One Health (in humans, animals, food, and environment) that will strengthen knowledge and evidence.
- Antimicrobial susceptibility mapping of in order to predict the trends in AMR.
- Formulating Policies for Infection Control in Healthcare settings, Antibiotic Management, Communities for control of AMR etc to Optimize the use of antimicrobial agents in all sectors; participate in “Swachh Bharat Abhiyan” and the “Kayakalp Programme”.
- Promote research and innovations for AMR activities by advising on Development of novel antimicrobials in addressing the challenge of AMR, and the Development of Diagnostic tools in addressing the challenge of AMR.
- Collaborate on AMR at international, national, and sub-national levels as a Partner in knowledge, action, and Solutions.
- Promote holistic approach by adopting the principles of AYUSH.
- Stewardship to improve awareness and understanding of AMR through effective communication, education, and training.
- Dr. Maneesh Paul is the AMR Action Steward since 2010 participating in Antibiotic Action initiated by Dr. Laura Piddock, AMR Committee of IDSA, AMR Cell of Govt. of Karnataka (India), and Expert on GARDP’s REVIVE program.
- AMRACE, short for Anti-Microbial Resistance Action Center of Excellence, is an innovative initiative led by Microvioma in collaboration with Bangalore Bioinnovation Center, aiming to address antimicrobial resistance challenges. Through a holistic approach focusing on Stewardship, Diagnostics, and Interventions, AMRACE empowers scientists and policymakers in India to combat microbial drug resistance effectively. By fostering collaborations with global Knowledge Partners and engaging with academic and non-academic institutions, AMRACE conducts practical activities and interventions, focusing on surveillance, diagnostics, immunotherapies, drug design, and policy formulation.